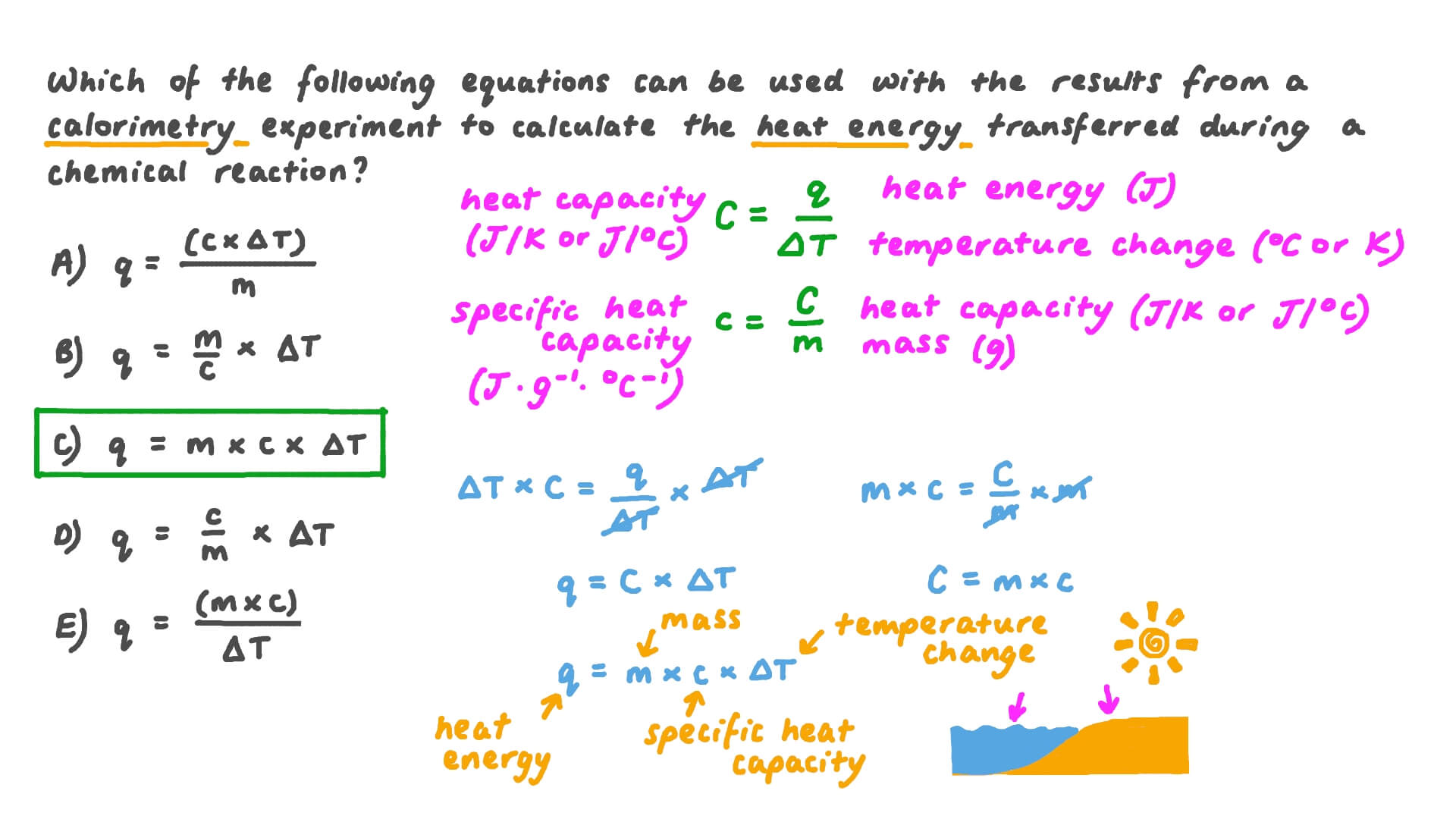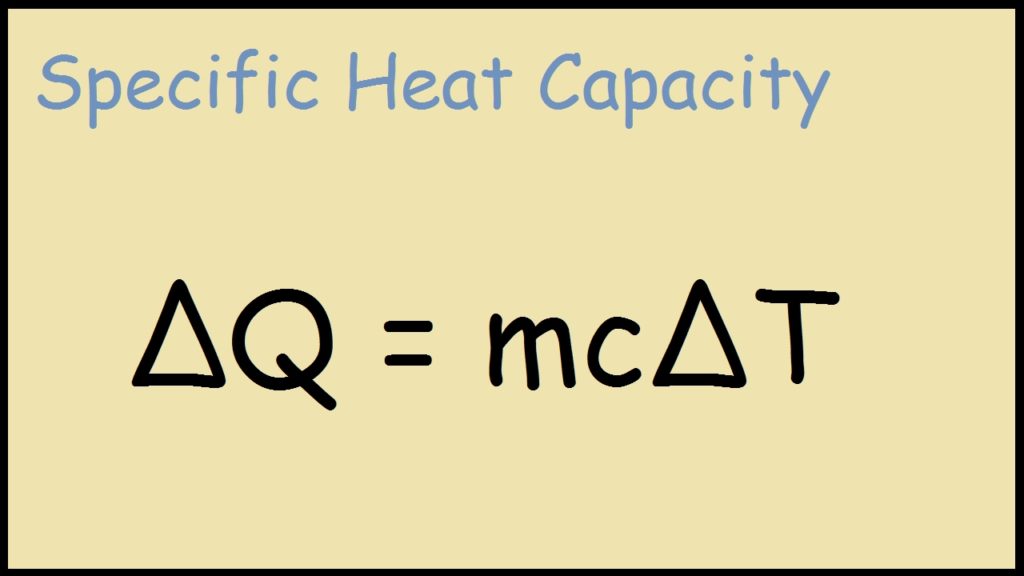Calculate the amount of heat required to raise the temperature of 13.5 g aluminium from 300 K to 400 K, if specific heat of aluminium is 0.9 J (.^(@)C)^(-1) g^(-1).

How to determine the heat evolved or consumed by the reaction of 1.0 g SO2(g) with excess oxygen, with results from a Hess' Law Equation? | Socratic